#Calculate cut off wavelength draw absorbtion spectrum software
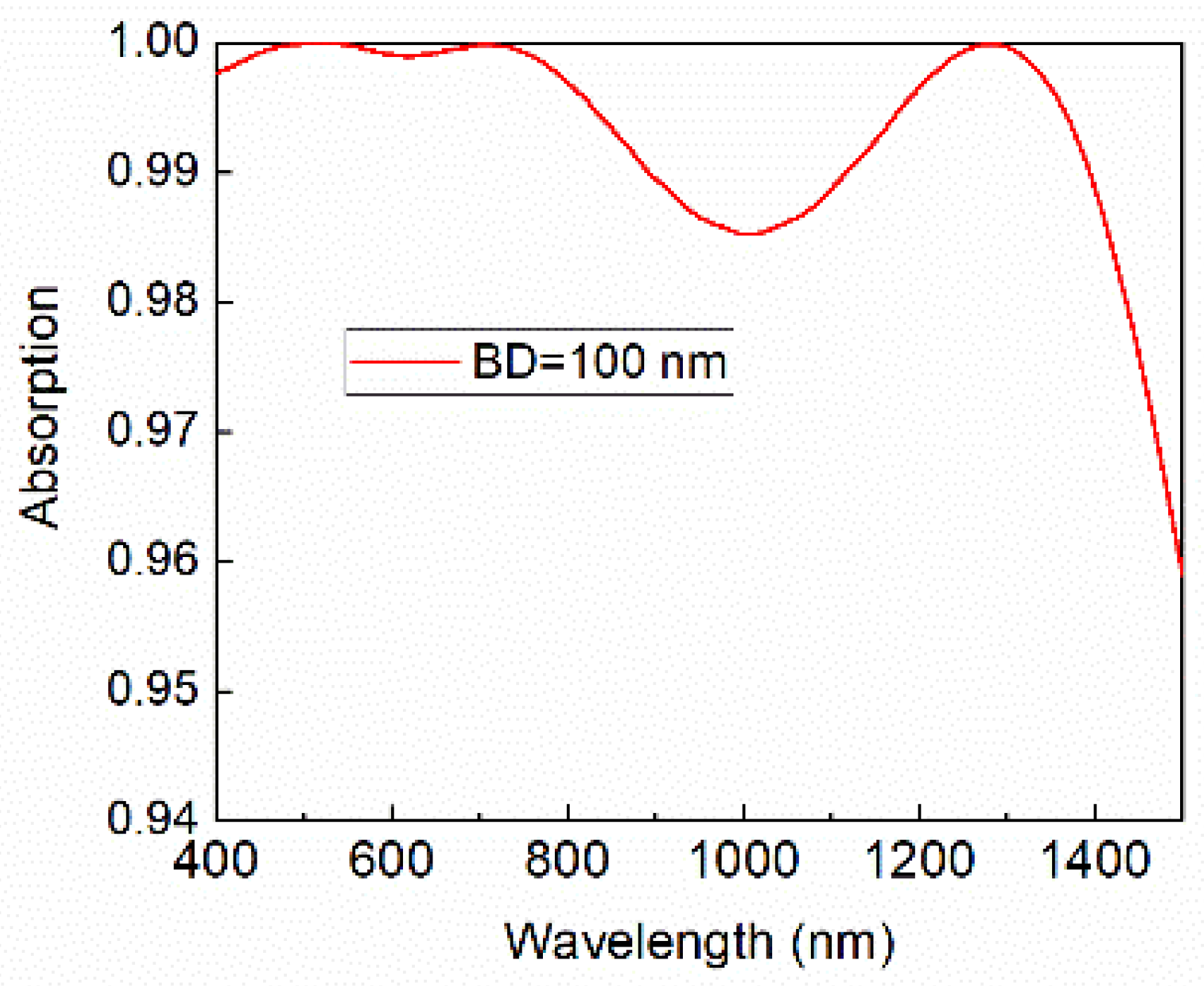
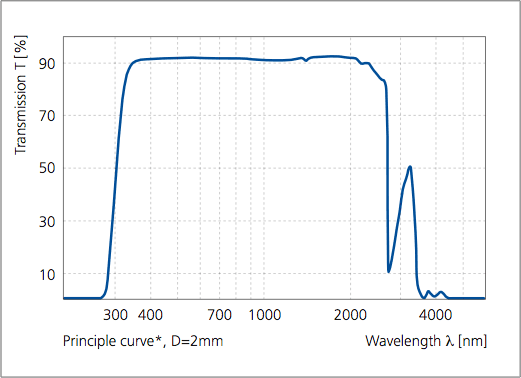
Commercial spreadsheet software was used to calculate normal emittance. Calculation of the visible transmittance, visible reflectance, solar transmittance, and solar reflectance was conducted using the solar transmittance measurement software shown in Fig. Table 2 shows the visible transmittance, visible reflectance, solar transmittance, solar reflectance, and normal emittance of each calculated sample. The low range of the color explains why it is difficult to distinguish this color in the spectral band. In the above color spectrum chart, indigo is made a subset of violet color. The entry unit of frequency can be modified, the output wavelength is calculated in meters. The frequency of wavelength range for indigo is around 425-450 nm and frequency of 670-700 THz. It assumes that the wave is traveling at the speed of light which is the case for most wireless signals. 2, 3 and 4 that large differences in both transmittance and reflectance exist in the UVVIS- NIR region, but the reflectance values in the infrared region were about the same. This frequency to wavelength calculator helps you determine the wavelength of a waveform based on the frequency. The measured samples consisted of 1 type of transparent glass and three types of opaque glass (green: transparent glass, black: opaque glass 1, red: opaque glass 2, blue: opaque glass 3). 2, 3 and 4 show the transmission spectra and reflection spectra in the UV-VIS-NIR region (correction by absolute reflectance of standard sample) and reflection spectra in the infrared region, respectively. 1 shows a glass sample placed on the integrating sphere. Absorption spectra are produced when white light, for example, is directed onto a particular substance and certain wavelengths in the spectrum of the white. Table 1 summarizes the measurement conditions that were used. This interaction time cannot be increased by lowering the light intensity.Measurements were conducted on four types of commercial plate glass to determine their respective visible transmittance, visible reflectance, solar transmittance, solar reflectance, and normal emittance values.
The absence of a lag time means that these one-on-one interactions occur instantaneously.
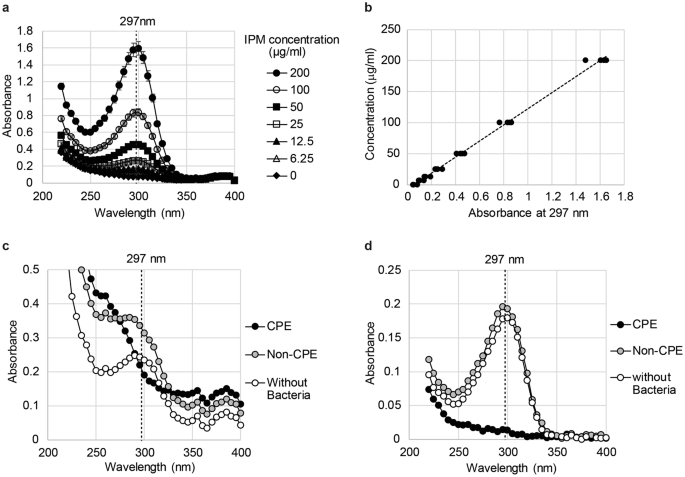
In Einstein’s interpretation, interactions take place between individual electrons and individual photons. The experimental setup to study the photoelectric effect is shown schematically in Figure \(\PageIndex\): Typical Values of the Work Function for Some Common Metals Metal Every atom of a certain element will have the same pattern of lines all the time. There are three parts of a spectrum: continuum emission (or blackbody radiation), emission lines, and absorption lines. Electrons that are emitted in this process are called photoelectrons. A plot of the brightness of an object versus wavelength is called a spectrum and can be observed using a tool called a spectroscope. Astronomical spectra can be combination of absorption and emission lines on. This phenomenon is known as the photoelectric effect. The visible spectrum, showing the wavelengths corresponding to each color. Polycrystalline nature of ZnO nanoparticle was observed in both ZnO powder samples prepared at pH 10.7. The particle diameter of ZnO nanoparticles calculated using the above equation was found to be 12.1256 nm. When a metal surface is exposed to a monochromatic electromagnetic wave of sufficiently short wavelength (or equivalently, above a threshold frequency), the incident radiation is absorbed and the exposed surface emits electrons. where, p is 396 nm (peak absorbance wavelength) and r represents radius of ZnO nanoparticles (Kumbhakar et al., 2008).

If you have the absorption spectrum of a solvent with one peak, your cut off wavelength will be the wavelength. Hydrogen Spectrum - Wavelength, Diagram, Hydrogen Emission Spectrum Hydrogen Spectrum introduction We all know that electrons in an atom or a molecule absorb energy and get excited, they jump from a lower energy level to a higher energy level, and they emit radiation when they come back to their original states.